Formidable Info About What Is An Example Of A Heat Transfer In Real Life
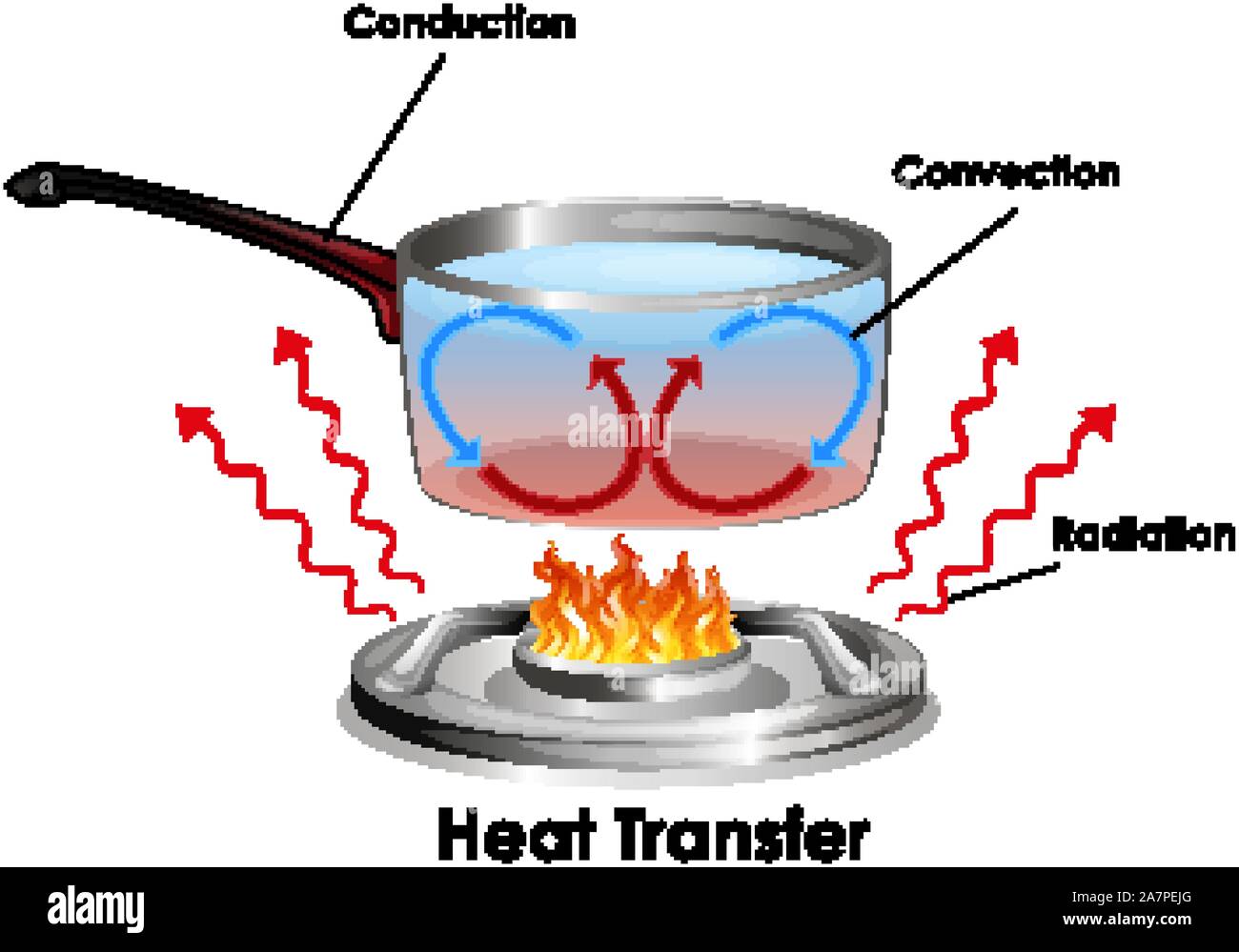
Heat Transfer All Around Us
1. Understanding the Basics of Heat Transfer
Ever wondered how your coffee cools down, or how a refrigerator manages to keep your leftovers from turning into a science experiment? The answer is heat transfer! It's a fundamental process that's happening all the time, everywhere, influencing everything from cooking to climate. Simply put, heat transfer is the movement of thermal energy from one place to another, driven by temperature differences. Think of it like a crowd of people: they naturally move from a crowded area to a less crowded one. Similarly, heat flows from a hotter object (where the molecules are buzzing with energy) to a cooler one (where the molecules are more relaxed).
There are actually three main ways heat can travel: conduction, convection, and radiation. Each method has its own quirks and favorite scenarios. And honestly, sometimes they all work together! Imagine a pot of soup on the stove — conduction heats the pot itself, convection circulates the broth, and radiation warms your face as you lean in for a sniff.
So, the keyword here is "heat transfer," and in this context, it's acting as a noun. It's the thing we're talking about: the actual process of thermal energy moving. We'll dive into some seriously relatable, real-life examples of this noun in action!
Get ready to see the world in a whole new, thermally-aware way! From the humble toaster to the vastness of space, heat transfer is the unsung hero making it all happen.

A Toasty Example
2. How Heat Moves Through Solids
Let's start with something familiar: your trusty toaster. Conduction is the star of the show here. Conduction is all about heat transfer through direct contact. Think of it as a game of dominoes, but with molecules. When you turn on the toaster, the heating element gets hot. This hot element then touches the metal of the toaster, and the heat starts moving through the metal itself. The molecules in the hot element vibrate like crazy, bumping into the molecules in the metal. This energy is passed along, molecule by molecule, until the outside surface heats up which toasts your bread.
That's why the toaster gets warm to the touch (careful!). Heat is literally being conducted from the heating element, through the metal, to the surface. And if you touch a metal spoon that's been sitting in a hot cup of tea, you'll experience conduction firsthand. It's a silent, steady transfer of energy that's essential for many everyday appliances and processes. The material itself makes a big difference, too. Metals are usually excellent conductors (that's why they're used in pots and pans), while materials like wood or plastic are poor conductors (that's why they're used for handles!).
The rate of conduction depends on factors like the material's thermal conductivity, the temperature difference, and the thickness of the material. A thick metal plate will take longer to heat up than a thin one, and a material with high thermal conductivity (like copper) will conduct heat much faster than one with low conductivity (like wood). So next time you're making toast, remember the tiny molecular dominoes working tirelessly inside!
So, conduction is the silent hero making sure your toast is perfectly golden brown, ensuring the heat from the heating element reaches every nook and cranny of your bread. It's a molecular chain reaction with delicious results.
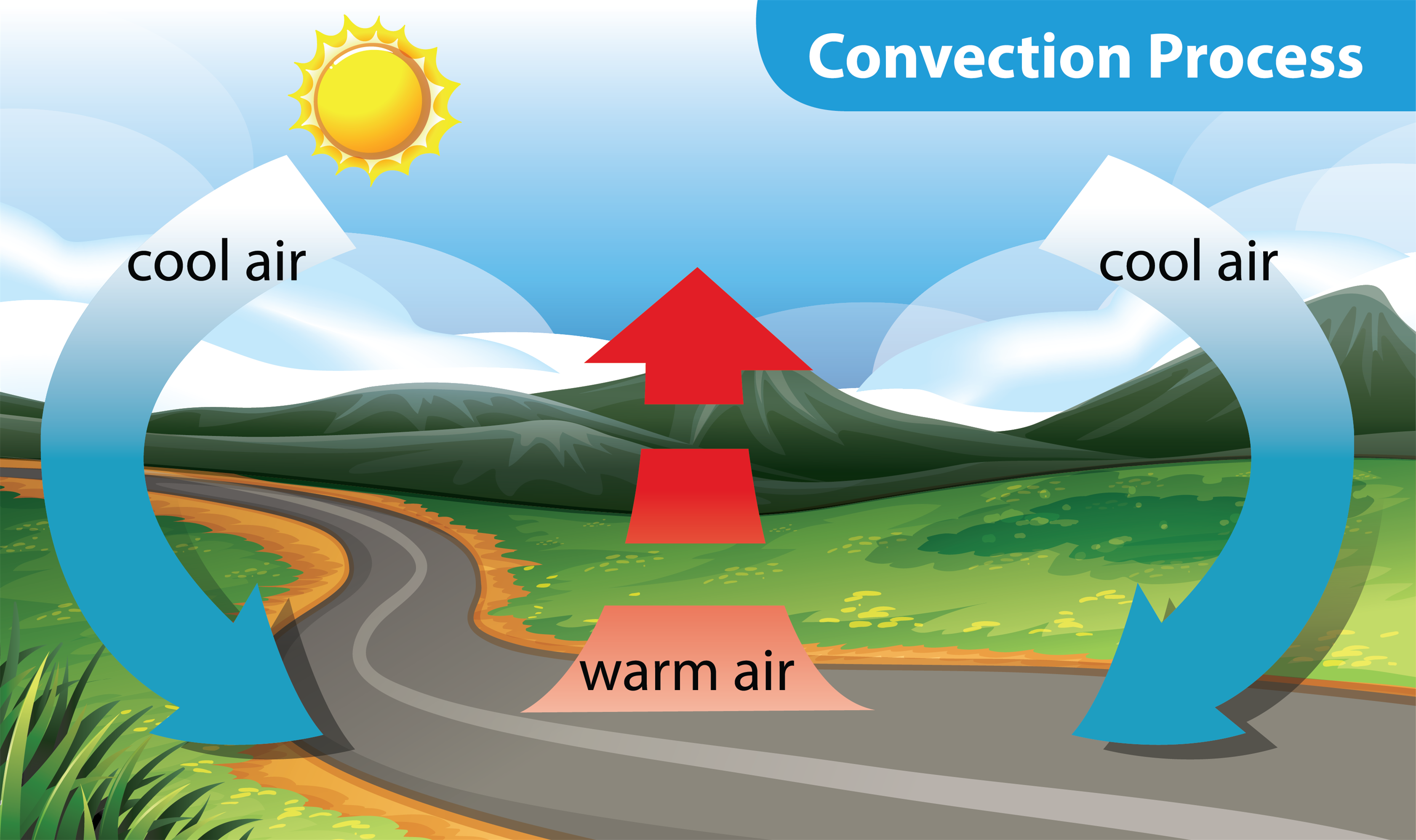
Boiling Water
3. Heat Transfer Through Fluids (Liquids and Gases)
Now, let's talk about boiling water — a classic example of convection. Convection is heat transfer through the movement of fluids (liquids or gases). When you heat a pot of water on the stove, the water at the bottom gets hot first. As it heats up, it becomes less dense and rises. This is because the molecules are moving faster and taking up more space. Colder, denser water then sinks to the bottom to replace the warmer water. This creates a circular flow, known as a convection current.
Think of it like a dance floor where the hottest dancers move to the front, pushing the cooler dancers to the back. This constant mixing ensures that the entire pot of water eventually reaches a uniform temperature. That's why bubbles form throughout the water, not just at the bottom. These bubbles are steam, created when the water gets hot enough to change from a liquid to a gas. The steam rises to the surface and escapes, taking heat with it.
Convection is responsible for many natural phenomena, such as weather patterns and ocean currents. Warm air rises, creating low pressure areas, while cold air sinks, creating high pressure areas. These pressure differences drive wind. Similarly, warm ocean currents, like the Gulf Stream, transport heat from the equator towards the poles, influencing the climate of many regions.
Even the air in your room is constantly circulating due to convection. A radiator or a heating vent heats the air around it, causing it to rise. Cooler air then sinks to replace the warmer air, creating a gentle flow that distributes heat throughout the room. Next time you're feeling a draft, remember that it's just convection doing its thing, mixing up the air and keeping things comfortable (or sometimes, uncomfortably chilly!).

Convection 20+ Examples, Definition, Formula, Types
Feeling the Sun
4. Heat Transfer Through Electromagnetic Waves
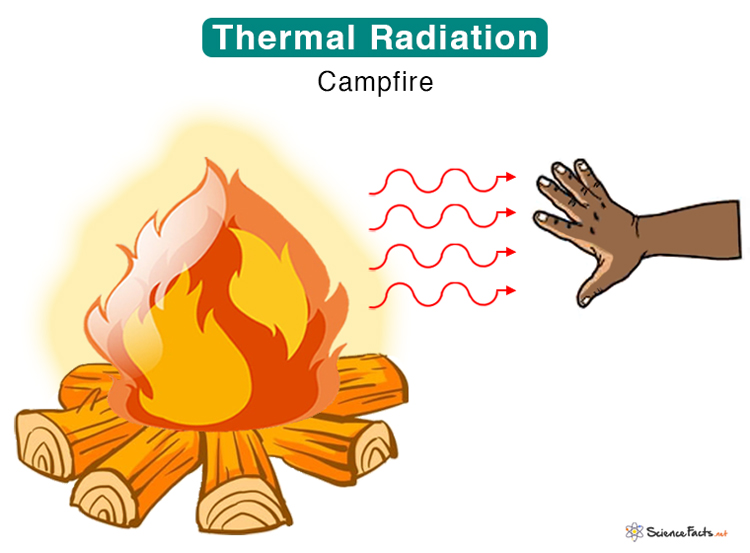
Finally, let's talk about radiation — the superhero of heat transfer because it doesn't need any medium at all! Radiation is heat transfer through electromagnetic waves. This is how the sun warms the Earth, even though there's a vast vacuum of space in between. Unlike conduction and convection, radiation doesn't require any physical contact or the movement of fluids. It's all about energy traveling in the form of electromagnetic waves, like light or infrared radiation. When these waves hit an object, they transfer their energy to the object, causing it to heat up.
That's why you can feel the warmth of a fire even if you're not standing right next to it. The fire is emitting infrared radiation, which travels through the air and warms your skin. Darker colors absorb more radiation than lighter colors, which is why wearing a black shirt on a sunny day can make you feel hotter. Similarly, greenhouses use glass to trap infrared radiation, creating a warmer environment for plants to grow. The glass allows sunlight to enter, but it prevents the infrared radiation emitted by the plants and soil from escaping.
Microwave ovens use a specific type of electromagnetic radiation to heat food. The microwaves penetrate the food and cause water molecules to vibrate, generating heat. This is a very efficient way to heat food quickly. It's also why some materials, like metal, can't be used in a microwave — they reflect the microwaves, which can damage the oven.
Even your own body is constantly radiating heat. That's why thermal cameras can be used to detect people in the dark. These cameras detect the infrared radiation emitted by the body and convert it into a visible image. Radiation is a powerful and versatile form of heat transfer that plays a vital role in many aspects of our lives, from keeping us warm on a sunny day to cooking our dinner.
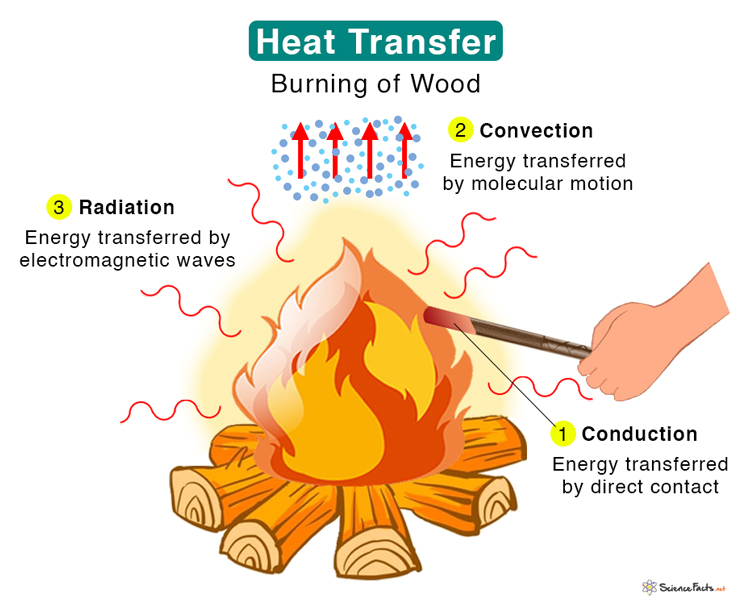
Combining All Three
5. A Symphony of Heat Transfer
Now, let's consider a campfire. A campfire isn't just one type of heat transfer in action; it's a beautiful symphony of all three! The logs themselves transfer heat through conduction. The flames heat the air around them, creating convection currents that warm you from a distance. And of course, the fire radiates warmth through radiation, allowing you to feel its heat even if you're not directly in the path of the rising hot air.
When you're roasting marshmallows, you're using all three methods. The metal skewer conducts heat to the marshmallow, the hot air convects around it, and the radiant heat from the embers toasts the outside to a perfect golden brown. The interplay of conduction, convection, and radiation is what makes a campfire such a comforting and effective source of heat and light.
Another example: Imagine a car parked in the sun. The sun's radiation heats the car's exterior. The metal of the car conducts this heat inwards, warming the interior surfaces. And finally, convection currents develop inside the car, circulating the hot air and making it feel like an oven! That's why it's always a good idea to crack a window to allow some of that hot air to escape.
Understanding how heat transfer works can help you make more informed decisions in your daily life. Whether you're choosing the right cookware, designing a more energy-efficient home, or simply trying to stay warm on a cold day, knowledge of conduction, convection, and radiation can be a valuable asset. So, the next time you feel the warmth of a fire or the coolness of a breeze, remember the amazing physics that's happening all around you.
FAQs About Heat Transfer
6. Your Burning Questions Answered
Okay, so we've covered the basics. But maybe you're still scratching your head about a few things. Let's tackle some frequently asked questions about heat transfer.
Q: What's the best way to insulate my house?
A: Insulation works by reducing heat transfer, primarily through conduction and convection. Good insulation materials, like fiberglass or foam, have low thermal conductivity, meaning they don't easily conduct heat. They also trap air, which further reduces convection. Proper insulation in your walls, attic, and floors can significantly reduce heat loss in the winter and heat gain in the summer, saving you money on your energy bills.
Q: Why does metal feel colder than wood, even if they're at the same temperature?
A: This is a classic example of conduction. Metal is a much better conductor of heat than wood. When you touch metal, it quickly draws heat away from your skin, making it feel cold. Wood, on the other hand, doesn't conduct heat as efficiently, so it doesn't draw heat away from your skin as quickly, making it feel warmer.
Q: How does a thermos keep liquids hot or cold?
A: A thermos uses a combination of strategies to minimize all three types of heat transfer. It typically has a double-walled construction with a vacuum between the walls. The vacuum eliminates conduction and convection because there's no medium for heat to travel through. The inner surfaces of the thermos are also often coated with a reflective material, which reduces heat transfer through radiation by reflecting the infrared radiation back into the liquid.
Q: Is heat transfer important in space?
A: Absolutely! In space, radiation is the primary mode of heat transfer, since there's no air or other medium for conduction or convection. Satellites and spacecraft are carefully designed to manage heat transfer through radiation. They often have reflective surfaces to minimize heat absorption from the sun and radiating fins to dissipate heat into space. Precise heat management is crucial for the proper functioning of electronic components and the survival of astronauts.